Search by medicine name. Napa OR Alatrol OR Amodis
Salost 5mg
TabletRisedronate Sodium
Unimed Unihealth MFG. Ltd
Other Strength:
- Salost 35mg
- Salost 150mg
Alternative:
Salost
Presentation
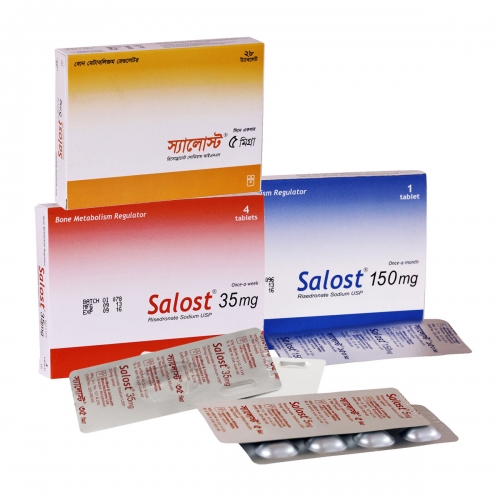
Salost 5mg tablet: Yellow, capsule shaped, film coated tablet; each tablet contains Risedronate Sodium hemi-pentahydrate USP equivalent to Risedronate Sodium 5mg.
Salost DR 35mg tablet: White, oval shaped, delayed release tablet; each tablet contains Risedronate Sodium hemi-pentahydrate USP equivalent to Risedronate Sodium 35mg.
Salost 150mg tablet: Blue, oval shaped, film coated tablet; each tablet contains Risedronate Sodium hemi-pentahydrate USP equivalent to Risedronate Sodium 150mg.
Indications
Treatment of postmenopausal osteoporosis to reduce the risk of vertebral fractures. Treatment of established postmenopausal osteoporosis to reduce the risk of hip fractures. Prevention of osteoporosis in postmenopausal women with increased risk of osteoporosis. To maintain or increase bone mass in postmenopausal women undergoing long-term (more than 3 months), systemic corticosteroid treatment at doses 7.5mg/day prednisone or equivalent. Treatment of osteoporosis in men at high risk of fractures.
Dosage and administration
Treatment of postmenopausal osteoporosis to reduce the risk of vertebral fractures and treatment of established postmenopausal osteoporosis, to reduce the risk of hip fractures: The recommended dose is Risedronate Sodium 5mg (one Salost 5mg tablet) once daily or Risedronate Sodium 35mg (one Salost DR 35mg tablet) once weekly on the same day each week or Risedronate Sodium 150mg (one Salost 150mg tablet) once monthly.
Prevention of osteoporosis in postmenopausal women with increased risk of osteoporosis: The recommended dose is Risedronate Sodium 5mg (one Salost 5mg tablet) once daily.
To maintain or increase bone mass in postmenopausal women undergoing long-term (more than 3 months), systemic corticosteroid treatment at doses 7.5mg/day prednisone or equivalent: The recommended dose is Risedronate Sodium 5mg (one Salost 5mg tablet) once daily.
Treatment of osteoporosis in men at high risk of fractures: The recommended dose is Risedronate Sodium 35mg (one Salost DR 35mg tablet) once weekly. The tablet should be taken on the same day each week.
Method of administration: The absorption of Risedronate Sodium film coated tablets are affected by food, thus to ensure adequate absorption patients should take Salost (Risedronate Sodium film coated) 5mg & 150mg tablets at least 30 minutes before the first food, other medicinal product or drink (other than plain water) of the day.
Salost (Risedronate Sodium film coated) 5mg & 150mg tablet: In the particular instances that before breakfast dosing is not practical, Salost (Risedronate Sodium film coated) 5mg tablet can be taken between meals or in the evening at the same time everyday, with strict adherence to the following instructions, to ensure Risedronate Sodium film coated tablet is taken on an empty stomach- Between meals: Risedronate Sodium film coated tablet should be taken at least 2 hours before and at least 2 hours after any food, medicinal product or drink (other than plain water). In the evening: Risedronate Sodium film coated tablet should be taken at least 2 hours after the last food, medicinal product or drink (other than plain water) of the day.
Salost DR (Risedronate Sodium delayed release) 35mg tablet: Should be taken in the morning, with breakfast, (this may include high fat foods, coffee, tea, milk, orange juice, etc. Salost DR (Risedronate Sodium delayed release) 35mg tablets should not be chewed, cut, or crushed. Care should be taken not to break the outer coating which is designed to remain intact until the tablet reaches the small intestine where the tablet coating dissolves and release the active ingredient.
If a dose is missed: Salost (Risedronate Sodium film coated) 5mg tablet: If an occasional dose is missed, Salost (Risedronate Sodium film coated) 5mg tablet can be taken before breakfast, between meals, or in the evening according to the instructions above.
Salost DR (Risedronate Sodium delayed release) 35mg tablet: Patients should be instructed that if a dose is missed, just take one Salost DR (Risedronate Sodium delayed release) 35mg tablet on the morning after remember. Patients should then return to taking one tablet once a week on the day the tablet is normally taken. Two tablets should not be taken on the same day.
Salost (Risedronate Sodium film coated) 150mg tablet: Patient should be instructed that if a dose is missed and the next month’s schedule dose is more than 7 days away, then should take the missed tablet in the morning after the day it is remembered. Patients should then return to taking one tablet once a month normally taken.
Salost (Risedronate Sodium film coated) 5mg, Salost DR (Risedronate Sodium delayed release) 35mg & Salost (Risedronate Sodium film coated) 150mg tablets must be swallowed whole and not sucked or chewed. To aid delivery of the tablet to the stomach Salost (Risedronate Sodium film coated) 5mg, Salost DR (Risedronate Sodium delayed release) 35mg & Salost (Risedronate Sodium film coated) 150mg tablets are to be taken while in an upright position with a glass of plain water (120ml or more). Patients should not lie down for 30 minutes after taking the tablet. Supplemental calcium and vitamin D should be considered if the dietary intake is inadequate.
Elderly: No dosage adjustment is necessary since bioavailability, distribution and elimination were similar in elderly (60 years of age or older) compared to younger subjects. This has also been shown in the very elderly, 75 years old and above postmenopausal population.
Renal impairment: No dosage adjustment is required for those patients with mild to moderate renal impairment. The use of Risedronate Sodium is contra-indicated in patients with severe renal impairment (Creatinine clearance lower than 30ml/min).
Contra-indications, warnings, etc
Contra-indications: Risedronate Sodium tablet is contra-indicated in patients
with known hypersensitivity to Risedronate Sodium and in patients with hypocalcaemia, severe renal impairment (creatinine clearance lower than 30ml/min), during pregnancy and lactation.
Precautions: Foods, drinks (other than plain water) and medicinal products containing polyvalent cations (such as calcium, magnesium, iron and aluminium) interfere with the absorption of bisphosphonates and should not be taken at the same time as Risedronate Sodium. In order to achieve the intended efficacy, strict adherence to dosing recommendations is necessary. Efficacy of bisphosphonates in the treatment of osteoporosis is related to the presence of low bone mineral density and/or prevalent fracture. High age or clinical risk factors for fracture alone are not sufficient reasons to initiate treatment of osteoporosis with a bisphosphonate. The evidence to support efficacy of bisphosphonates including Risedronate in the very elderly (>80 years) is limited. Bisphosphonates have been associated with oesophagitis, gastritis, oesophageal ulcerations and gastroduodenal ulcerations. Thus, caution should be used in patients who have a history of oesophageal disorders which delay oesophageal transit or emptying (e.g. stricture or achalasia) or in patients who are unable to stay in the upright position for at least 30 minutes after taking the tablet or if Risedronate is given to patients with active or recent oesophageal or upper gastrointestinal problems. Prescribers should emphasize to patients the importance of paying attention to the dosing instructions and be alert to any signs and symptoms of possible oesophageal reaction. The patients should be instructed to seek timely medical attention if they develop symptoms of oesophageal irritation such as dysphagia, pain on
swallowing, retrosternal pain or new/worsened heartburn. Hypocalcaemia should be treated before starting Risedronate Sodium therapy. Other disturbances of bone and mineral metabolism (i.e. parathyroid dysfunction, hypovitaminosis D) should be treated at the time of starting Risedronate Sodium therapy. Osteonecrosis of the jaw, generally associated with tooth extraction and/or local infection (including osteomyelitis) has been reported in patients with cancer receiving treatment regimens including primarily intravenously administered bisphophonates. Many of these patients were also receiving chemotherapy and corticosteroids. Osteonecrosis of the jaw has also been reported in patients with osteoporosis receiving oral bisphosphonates. A dental examination with appropriate preventive dentistry should be considered prior to treatment with bisphosphonates in patients with concomitant risk factors (e.g. cancer, chemotherapy, radiotherapy, corticosteroids, poor oral hygiene). While on treatment, these patients should avoid invasive dental procedures if possible. For patients who develop osteonecrosis of the jaw while on bisphosphonate therapy, dental surgery may exacerbate the condition. For patients requiring dental procedures, there are no data available to suggest whether discontinuation of bisphosphonate treatment reduces the risk of osteonecrosis of the jaw. Clinical judgment of the treating physician should guide the management plan of each patient based on individual benefit /risk assessment.
Use in pregnancy and lactation: There are no adequate data from the use of Risedronate Sodium in pregnant women. Studies in animals have shown reproductive toxicity. The potential risk for humans is unknown. Studies in animal indicate that a small amount of Risedronate Sodium pass into breast milk. Risedronate Sodium must not be used during pregnancy or by breast-feeding women.
Pediatric use: Safety and efficacy of Risedronate Sodium tablet have not been established in children and adolescents.
Side effects: Common: headache, constipation, dyspepsia, nausea, abdominal pain, diarrhoea, musculoskeletal pain. Uncommon: gastritis, oesophagitis, dysphasia, duodenitis, oesophageal ulcer. Rare: glossitis, oesophageal stricture.
Drug interactions: No formal interaction studies have been performed, however no clinically relevant interactions with other medicinal products were found during clinical trials. Among regular acetyl salicylic acid or NSAID users (3 or more days per week) the incidence of upper gastrointestinal adverse events in Risedronate Sodium tablet treated patients was similar to that in control patients. If considered appropriate Risedronate Sodium may be used concomitantly with oestrogen supplementation (for women only). Concomitant ingestion of medications containing polyvalent cations (e.g. calcium, magnesium, iron and aluminium) will interfere with the absorption of Risedronate Sodium. Risedronate Sodium is not systemically metabolized, does not induce cytochrome P450 enzymes, and has low protein binding.
Overdose: No specific information is available on the treatment of overdose with Risedronate Sodium. Decreases in serum calcium following substantial overdose may be expected. Signs
and symptoms of hypocalcaemia may also occur in some of these patients. Milk or antacids containing magnesium, calcium or aluminium should be given to bind Risedronate and reduce absorption of Risedronate Sodium. In cases of substantial overdose, gastric lavage may be considered to remove unabsorbed Risedronate Sodium.
Pharmaceutical precautions
Store in a cool and dry place protected from light.
Packaging quantities
Salost 5mg tablet: Carton containing 28 tablets in alu-alu blister.
Salost DR 35mg tablet: Carton containing 4 tablets in alu-alu blister.
Salost 150mg tablet: Carton containing 1 tablet in alu-alu blister.
এই পাতাটি ৫৩৩ বার দেখা হয়েছে
